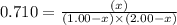Consider the reaction, which takes place at a certain elevated temperature CO(g)+NH3(g)⇌HCONH2(g), Kc=0.710 If a reaction vessel initially contains only CO and NH3 at concentrations of 1.00 M and 2.00 M, respectively, what will the concentration of HCONH2 be at equilibrium?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2


Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2
You know the right answer?
Consider the reaction, which takes place at a certain elevated temperature CO(g)+NH3(g)⇌HCONH2(g), K...
Questions

Mathematics, 27.10.2019 01:43


Mathematics, 27.10.2019 01:43









Mathematics, 27.10.2019 01:43





Mathematics, 27.10.2019 01:43

Mathematics, 27.10.2019 01:43


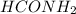 at equilibrium is, 0.513 M
at equilibrium is, 0.513 M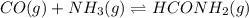
![K_c=\frac{[HCONH_2]}{[CO][NH_3]}](/tpl/images/0539/2269/5249e.png)