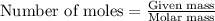Problem Page Question An analytical chemist weighs out of an unknown monoprotic acid into a volumetric flask and dilutes to the mark with distilled water. He then titrated this solution with solution. When the titration reaches the equivalence point, the chemist finds he has added of solution. Calculate the molar mass of the unknown acid. Round your answer to significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 3

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 12:30
Which element has the lowest electronegativity? calcium(ca) gallium(ga) selenium(se) bromine(br)
Answers: 1
You know the right answer?
Problem Page Question An analytical chemist weighs out of an unknown monoprotic acid into a volumetr...
Questions

Mathematics, 04.07.2019 23:00

Mathematics, 04.07.2019 23:00




Business, 04.07.2019 23:00

History, 04.07.2019 23:00



Mathematics, 04.07.2019 23:00

Mathematics, 04.07.2019 23:00



Social Studies, 04.07.2019 23:00

Biology, 04.07.2019 23:00



Biology, 04.07.2019 23:00

Mathematics, 04.07.2019 23:00

Social Studies, 04.07.2019 23:00

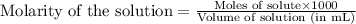
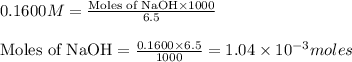

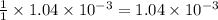 moles of HA
moles of HA