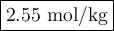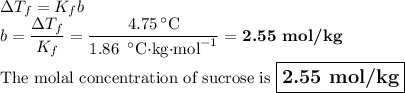Chemistry, 09.03.2020 06:03 kyleee1991
A solution of water (Kf=1.86 ∘C/m) and glucose freezes at − 4.75 ∘C. What is the molal concentration of glucose in this solution? Assume that the freezing point of pure water is 0.00 ∘C.

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
A solution of water (Kf=1.86 ∘C/m) and glucose freezes at − 4.75 ∘C. What is the molal concentration...
Questions

English, 21.09.2021 09:00

English, 21.09.2021 09:00



Mathematics, 21.09.2021 09:00

English, 21.09.2021 09:00

Mathematics, 21.09.2021 09:00



Mathematics, 21.09.2021 09:00

Biology, 21.09.2021 09:00



Mathematics, 21.09.2021 09:00

Chemistry, 21.09.2021 09:00

History, 21.09.2021 09:00



Mathematics, 21.09.2021 09:00