
Chemistry, 07.03.2020 07:31 GabbyLandry
The molar mass of a compound is 92 g/mol. Analysis of a sample of the compound indicates that it contains 0.606g of N and 1.390g of O. Find its molecular formula.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 23.06.2019 00:30
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2
You know the right answer?
The molar mass of a compound is 92 g/mol. Analysis of a sample of the compound indicates that it con...
Questions


Biology, 23.10.2019 03:20

Mathematics, 23.10.2019 03:20

Social Studies, 23.10.2019 03:20

History, 23.10.2019 03:20

Mathematics, 23.10.2019 03:30




Chemistry, 23.10.2019 03:30

Computers and Technology, 23.10.2019 03:30

Mathematics, 23.10.2019 03:30



History, 23.10.2019 03:30

English, 23.10.2019 03:30


English, 23.10.2019 03:30


Mathematics, 23.10.2019 03:30

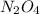
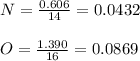
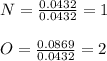
![[NO2]n = 92[14 + (16x2)]n = 92[14 +32]n = 9246n = 92](/tpl/images/0538/1065/5116b.png)
![[NO2]n \\= [NO2]2\\ = N2O4](/tpl/images/0538/1065/6f423.png)


