
Chemistry, 07.03.2020 05:36 damien1030
Calculate the pressures of NO, Cl2, and NOCl in an equilibrium mixture produced by the reaction of a starting mixture with 8.2 atm NO and 4.1 atm Cl2. (Hint: Kp is relatively large; assume the reaction goes to completion then comes back to equilibrium.)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:40
For a patient with the following pes statement and interventions, which would be the most appropriate monitoring and evaluating data? pes statement: inadequate calcium intake related to food and nutrition related knowledge deficit as evidenced by statements that the only dietary source of calcium is milk and she believes that she is lactose intolerant. patient’s nutrition prescription is for a diet providing 1200 mg calcium per day. patient was provided with in-depth nutrition education on alternative dietary and supplement sources of calcium. a. calcium intake (at subsequent visit) b. knowledge assessment by asking patient to identify food sources from menus and shopping list (at the end of the current visit) c. serum calcium (at next visit) d. both a and b e. both a and c
Answers: 2

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1
You know the right answer?
Calculate the pressures of NO, Cl2, and NOCl in an equilibrium mixture produced by the reaction of a...
Questions

Mathematics, 25.06.2019 23:00



History, 25.06.2019 23:00











History, 25.06.2019 23:00



Spanish, 25.06.2019 23:00


Biology, 25.06.2019 23:00

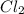 , and NOCl in an equilibrium mixture produced by the reaction of a starting mixture with 8.2 atm NO and 4.1 atm
, and NOCl in an equilibrium mixture produced by the reaction of a starting mixture with 8.2 atm NO and 4.1 atm 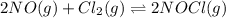
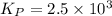
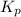 of the reaction is as follows.
of the reaction is as follows.![K_{P} = \frac{[NOCl]^{2}}{[NO]^{2}[Cl_{2}]}](/tpl/images/0537/9927/829f4.png)
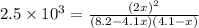
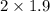 = 3.8
= 3.8

