
Chemistry, 07.03.2020 05:06 cnfndbxbfbdb2031
You have an evacuated container of fixed volume and known mass and introduce a known mass of a gas sample. measuring the pressure at constant temperature over time, you are surprised to see it slowly dropping. you measure the mass of the gas-filled container and find that the mass is what it should be - gas plus container - and the mass does not change over time, so you do not have a leak. part a suggest an explanation for your observations. suggest an explanation for your observations.
1. the gas undergoes a chemical reaction that has fewer gas particles in products than in reactants. pressure is directly proportional to number of particles, so the pressure decreased.
2. when the gas was plased into container it obtained some extra kinetic energy after some time this energy was expend, so the speed of the particles reduced and the pressure dropped the gas
3. undergoes a chemical reaction that has more gas particles in products than in reactants.
4. pressure is directly proportional to number of mass.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3

Chemistry, 23.06.2019 03:30
Mr. rose asked his student to draw a quadrilateral with four unequal sides. an example of this kind of quadrilateral
Answers: 1

Chemistry, 23.06.2019 07:00
Why do the strengths of london (dispersion) forces generally increase with increasing molecular size? choose one: a. heavier atoms have stronger attractions for each other than lighter atoms. b. dispersion forces are all equal in magnitude; there is no size dependence. c. dispersion forces arise from the attraction between the nuclei of atoms, and larger molecules have larger nuclei. d. dispersion forces arise from dipoles caused by the electron distribution being distorted. larger molecules have more electrons and, therefore, more distortions and a bigger force. e. dispersion forces depend on distance. larger molecules are farther apart and so the forces are smaller.
Answers: 2
You know the right answer?
You have an evacuated container of fixed volume and known mass and introduce a known mass of a gas s...
Questions








Geography, 11.07.2019 08:00

Geography, 11.07.2019 08:00




Mathematics, 11.07.2019 08:00

Mathematics, 11.07.2019 08:00



History, 11.07.2019 08:00

History, 11.07.2019 08:00

Mathematics, 11.07.2019 08:00

Mathematics, 11.07.2019 08:00

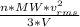
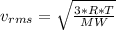 as such for pressure depends only on the number of moles so option 2 is cancelled out
as such for pressure depends only on the number of moles so option 2 is cancelled out

