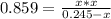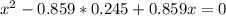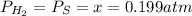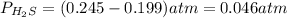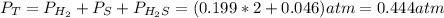Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
The isotonic saline solution described in part a is connected to an unknown solution via a semipermeable membrane, the unknown solution level drops. based on this information, what can be said about these two solutions?
Answers: 1


Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3
You know the right answer?
At a certain temperature, the K p for the decomposition of H 2 S is 0.859 . H 2 S ( g ) − ⇀ ↽ − H 2...
Questions

Mathematics, 11.05.2021 04:00

Biology, 11.05.2021 04:00




Spanish, 11.05.2021 04:00



Mathematics, 11.05.2021 04:00

History, 11.05.2021 04:00



Mathematics, 11.05.2021 04:00

History, 11.05.2021 04:00

Mathematics, 11.05.2021 04:00

Mathematics, 11.05.2021 04:00




Mathematics, 11.05.2021 04:00

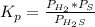 (1)
(1)