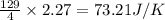Chemistry, 07.03.2020 00:46 flowerrbabie
Consider the reaction 4HCl(g) + O2(g)2H2O(g) + 2Cl2(g) Using standard thermodynamic data at 298K, calculate the entropy change for the surroundings when 2.27 moles of HCl(g) react at standard conditions. Ssurroundings = J/K

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:00
What layer of the atmosphere is directly above the troposphere?
Answers: 1

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 10:00
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
Consider the reaction 4HCl(g) + O2(g)2H2O(g) + 2Cl2(g) Using standard thermodynamic data at 298K, ca...
Questions


Computers and Technology, 22.02.2020 04:31


Biology, 22.02.2020 04:31











Biology, 22.02.2020 04:32




English, 22.02.2020 04:32

Mathematics, 22.02.2020 04:32

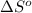 for the surrounding when given amount of HCl gas is reacted is 73.21 J/K
for the surrounding when given amount of HCl gas is reacted is 73.21 J/K![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0536/8507/52737.png)
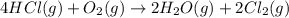
![\Delta S^o_{rxn}=[(2\times \Delta S^o_{(Cl_2(g))})+(2\times \Delta S^o_{(H_2O(g))})]-[(4\times \Delta S^o_{(HCl(g))})+(1\times \Delta S^o_{(O_2(g))})]](/tpl/images/0536/8507/2475f.png)
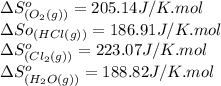
![\Delta S^o_{rxn}=[(2\times (223.07))+(2\times (188.82))]-[(4\times (186.91))+(1\times (205.14))]\\\\\Delta S^o_{rxn}=-129J/K](/tpl/images/0536/8507/8e27e.png)