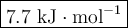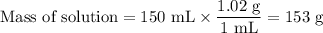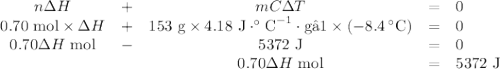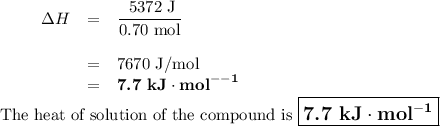0.70 moles of an unknown solid is placed into water to make 150.0 mL of solution. The solution's temperature decreases by 8.4°C. Calculate ∆H for the dissolution of the unknown solid. (The specific heat of the solution is 4.18 J/g・°C and the density of the solution is 1.02 g/mL).

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
2h2s + 3o2 2so2 + 2h2o which option gives the correct mole ratios? h2s: so2 = 2: 2 and o2: h2o = 3: 2 h2s: so2 = 2: 3 and o2: h2o = 3: 2 h2s: so2 = 4: 4 and o2: h2o = 5: 4 h2s: so2 = 4: 6 and o2: h2o = 4: 4
Answers: 1

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3

Chemistry, 22.06.2019 16:00
As changes in energy levels of electrons increase, the frequencies of atomic line spectra they emit
Answers: 2

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2
You know the right answer?
0.70 moles of an unknown solid is placed into water to make 150.0 mL of solution. The solution's tem...
Questions



Mathematics, 01.02.2022 16:00

Mathematics, 01.02.2022 16:00



Mathematics, 01.02.2022 16:00

Mathematics, 01.02.2022 16:10

Mathematics, 01.02.2022 16:10

Social Studies, 01.02.2022 16:10

Mathematics, 01.02.2022 16:10

English, 01.02.2022 16:10

Mathematics, 01.02.2022 16:10




Computers and Technology, 01.02.2022 16:10


Social Studies, 01.02.2022 16:10