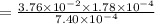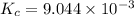Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 22.06.2019 19:30
Which liquid (h2o, h2o + soap, or h2o + salt) has the strongest cohesion and adhesion? (need now plz)
Answers: 1

Chemistry, 22.06.2019 21:30
Describe at least two advantages and two disadvantages of using hydropower as a source of energy.
Answers: 2
You know the right answer?
Some COCl2 is placed in a sealed flask and heated to 756 K. When equilibrium is reached, the flask i...
Questions

Biology, 30.01.2020 02:55






English, 30.01.2020 02:55

History, 30.01.2020 02:55



Biology, 30.01.2020 02:55

Biology, 30.01.2020 02:55

Mathematics, 30.01.2020 02:55



Mathematics, 30.01.2020 02:55

Social Studies, 30.01.2020 02:55


Mathematics, 30.01.2020 02:55

Business, 30.01.2020 02:55

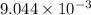 is the value of the equilibrium constant for this reaction at 756 K.
is the value of the equilibrium constant for this reaction at 756 K.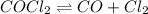
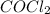
![[COCl_2]=7.40\times 10^{-4} M](/tpl/images/0534/1166/1134d.png)
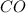
![[CO]=3.76\times 10^{-2} M](/tpl/images/0534/1166/484f5.png)
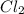
![[Cl_2]=1.78\times 10^{-4} M](/tpl/images/0534/1166/d4643.png)
![K_c=\frac{[CO][Cl_2]}{[COCl_2]}](/tpl/images/0534/1166/59c52.png)