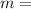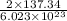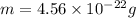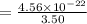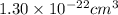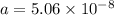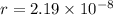Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 23.06.2019 01:00
Atoms contain subatomic particles called protons and neutrons. when these protons and neutrons spilt, a lot of energy is released
Answers: 3

Chemistry, 23.06.2019 13:30
Which of these statements describes the size of an atom? a. an atom is larger than a sheet of aluminum foil. b. an atom is small but can be seen with just our eyes. c. an atom is the size of a plastic building block. d. an atom is tiny and cannot be seen without magnification.
Answers: 2
You know the right answer?
The atoms in barium metal are arranged in a bodycentered cubic unit cell. Calculate the radius of a...
Questions







Mathematics, 11.02.2020 00:11




Business, 11.02.2020 00:11

History, 11.02.2020 00:11

Business, 11.02.2020 00:11




Mathematics, 11.02.2020 00:12

Computers and Technology, 11.02.2020 00:12


Biology, 11.02.2020 00:12

 10^-8
10^-8