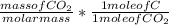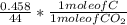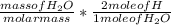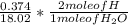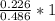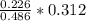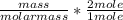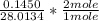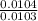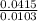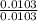Chemistry, 05.03.2020 00:53 hbhdnjdndj1867
Dimethylhydrazine is a carbon-hydrogen-nitrogen compound used in rocket fuels. When burned in an excess of oxygen, a 0.312 gg sample yields 0.458 gg CO2CO2 and 0.374 gg H2OH2O. The nitrogen content of a 0.486 gg sample is converted to 0.226 gg N2N2.What is the empirical formula of dimethylhydrazine?Express your answer as a chemical formula.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:00
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1


Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3

Chemistry, 23.06.2019 01:00
Iron (fe) reacts with copper sulfate (cuso4) to form iron (ii) sulfate. in this reaction, cu2+ gains electrons to form cu. which statement is true about this reaction? fe(s) + cuso4(aq) → feso4(aq) + cu(s)
Answers: 3
You know the right answer?
Dimethylhydrazine is a carbon-hydrogen-nitrogen compound used in rocket fuels. When burned in an exc...
Questions



Biology, 21.03.2020 06:08

Mathematics, 21.03.2020 06:08

History, 21.03.2020 06:08



Spanish, 21.03.2020 06:09

History, 21.03.2020 06:09




English, 21.03.2020 06:09



History, 21.03.2020 06:10


Computers and Technology, 21.03.2020 06:10


Mathematics, 21.03.2020 06:10