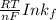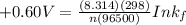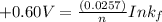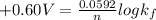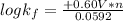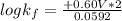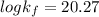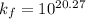Chemistry, 04.03.2020 22:59 emilyanneK236
Be sure to answer all parts. Zinc is an amphoteric metal, meaning it reacts with both acids and bases. The standard reduction potential is −1.36 V for the following reaction: (1)Zn(OH)42−(aq) + 2e− → Zn(s) + 4OH−(aq) Calculate the formation constant Kf for the reaction: (2)Zn2+(aq) + 4OH−(aq) ⇌ Zn(OH)42−(aq)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 23.06.2019 02:00
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3
You know the right answer?
Be sure to answer all parts. Zinc is an amphoteric metal, meaning it reacts with both acids and base...
Questions

Mathematics, 25.06.2019 09:10




Health, 25.06.2019 09:10


Spanish, 25.06.2019 09:10









Mathematics, 25.06.2019 09:10


Mathematics, 25.06.2019 09:10



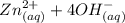 ⇄
⇄ 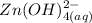

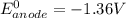

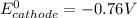
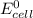 is given as:
is given as: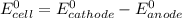
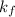 of the
of the