

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

Chemistry, 22.06.2019 12:30
Which element has the lowest electronegativity? calcium(ca) gallium(ga) selenium(se) bromine(br)
Answers: 1

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2
You know the right answer?
Consider the mechanism. Step 1: A + B − ⇀ ↽ − C equilibrium Step 2: C + A ⟶ D slow Overall: 2 A + B...
Questions


Chemistry, 11.11.2020 04:10


Business, 11.11.2020 04:10

Mathematics, 11.11.2020 04:10

Health, 11.11.2020 04:10



Mathematics, 11.11.2020 04:10





Social Studies, 11.11.2020 04:10

History, 11.11.2020 04:10

Chemistry, 11.11.2020 04:10




![Rate=k'[A]^2[B]](/tpl/images/0533/4284/3527f.png)
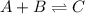 (fast)
(fast)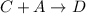 (slow)
(slow)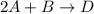
![Rate=k[C][A]](/tpl/images/0533/4284/5d5c1.png) ............(1)
............(1)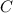 , we get:
, we get:![K=\frac{[C]}{[A][B]}](/tpl/images/0533/4284/f1e20.png)
![[C]=K\times [A][B]](/tpl/images/0533/4284/91a27.png) ...........(2)
...........(2)![Rate=k[A]\times K\times [A][B]](/tpl/images/0533/4284/847a2.png)
![Rate=K\times k[A]^2[B]](/tpl/images/0533/4284/fb0ae.png)


