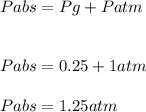Chemistry, 04.03.2020 03:27 akimadixon13
A ball has a volume of 5.27 liters and is at a temperature of 27.0°C. A pressure gauge attached to the ball reads 0.25 atmosphere. The atmospheric pressure is 1.00 atmosphere.
Calculate the absolute pressure inside the ball and the amount of air it contains.
The absolute pressure inside the ball is ??? atmospheres
The ball contains ???mole of air

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 10:00
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
You know the right answer?
A ball has a volume of 5.27 liters and is at a temperature of 27.0°C. A pressure gauge attached to t...
Questions

Physics, 20.10.2019 08:20




Mathematics, 20.10.2019 08:20

History, 20.10.2019 08:20

Spanish, 20.10.2019 08:20

Social Studies, 20.10.2019 08:20

Geography, 20.10.2019 08:20


Physics, 20.10.2019 08:30

Mathematics, 20.10.2019 08:30

Geography, 20.10.2019 08:30

History, 20.10.2019 08:30





History, 20.10.2019 08:30