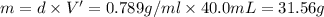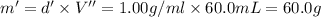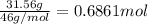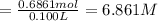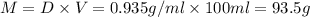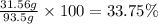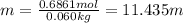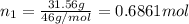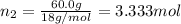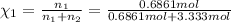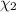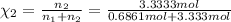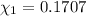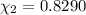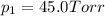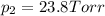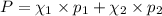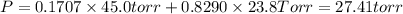Chemistry, 04.03.2020 02:22 bgallman153p71edg
Grey Goose ® vodka has an alcohol content of 40.0 % (v/v). Assuming that vodka is composed of only ethanol and water answer the following questions. Note: The molar masses of water and ethanol are 18.0 g and 46.0 g, respectively. The densities of water, ethanol, and this vodka mixture are 1.00 g/mL, 0.789 g/mL, and 0.935 g/mL, respectively
a. Calculate the molarity of ethanol in this vodka, assuming that water is the solvent.
b. Calculate the percent by mass of ethanol % (m/m) in this vodka.
c. Calculate the molality of ethanol in this vodka assuming that water is the solvent.
d. Calculate the mole fractions of ethanol and water in this vodka.
e. Calculate the vapor pressure, in torr, of this vodka at 25.0 oC if the vapor pressures of pure water and ethanol are 23.8 torr and 45.0 torr, respectively?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
How does a hydroelectric power plant converts energy into energy.
Answers: 1

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2

Chemistry, 22.06.2019 18:30
When a device is used in a circuit in which the voltage is 81 v the current flowing through the device is 3 a what is the resistance of the device
Answers: 2
You know the right answer?
Grey Goose ® vodka has an alcohol content of 40.0 % (v/v). Assuming that vodka is composed of only e...
Questions

History, 18.03.2021 18:10

Mathematics, 18.03.2021 18:10


English, 18.03.2021 18:10

History, 18.03.2021 18:10





Social Studies, 18.03.2021 18:10


Mathematics, 18.03.2021 18:10

World Languages, 18.03.2021 18:10


Business, 18.03.2021 18:10


History, 18.03.2021 18:10

Mathematics, 18.03.2021 18:10