
Chemistry, 03.03.2020 05:45 sarahalexa19
What is the total pressure (in atm) inside of a vessel containing N2 exerting a partial pressure of 0.256 atm, He exerting a partial pressure of 203 mmHg, and H2 exerting a partial pressure of 39.0 kPa?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 21:00
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1


Chemistry, 23.06.2019 01:00
Which of the following is a physical change? a.burning a piece of wood b.sawing a piece of wood in half c.rust forming on an iron fence d.a copper roof changing color from orange to green
Answers: 1
You know the right answer?
What is the total pressure (in atm) inside of a vessel containing N2 exerting a partial pressure of...
Questions

Biology, 12.05.2021 20:20


Mathematics, 12.05.2021 20:20


Social Studies, 12.05.2021 20:20

Mathematics, 12.05.2021 20:20

English, 12.05.2021 20:20


English, 12.05.2021 20:20

English, 12.05.2021 20:20

Mathematics, 12.05.2021 20:20

Mathematics, 12.05.2021 20:20


Geography, 12.05.2021 20:20



Computers and Technology, 12.05.2021 20:20



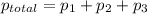
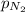 = partial pressure of nitrogen = 0.256 atm
= partial pressure of nitrogen = 0.256 atm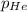 = partial pressure of helium = 203 mm Hg = 0.267 atm (760mmHg=1atm)
= partial pressure of helium = 203 mm Hg = 0.267 atm (760mmHg=1atm)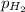 = partial pressure of hydrogen =39.0 kPa = 0.385 atm (1kPa=0.00987 atm)
= partial pressure of hydrogen =39.0 kPa = 0.385 atm (1kPa=0.00987 atm)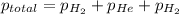
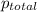 =0.256atm+0.267atm+0.385atm =0.908atm
=0.256atm+0.267atm+0.385atm =0.908atm

