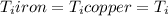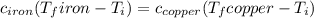Five-gram samples of copper and iron are at room temperature. both samples receive equal amounts of energy due to heat flow. the specific heat capacity of copper is 0.09 cal/g°c, and the specific heat capacity of iron is 0.11 cal/g°c. which of the following statements is true? the temperature of each sample will increase by the same amount. the temperature of each sample will decrease by the same amount. the copper will get hotter than the iron. the iron will get hotter than the copper.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Is this a scientific model? use complete sentences to explain why or why not. a graphic organizer showing the water cycle
Answers: 3

Chemistry, 21.06.2019 16:00
Review the branily terms and services guides well u know what i never did so go have a nice ice cream sunday
Answers: 1

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 15:30
Light waves can move through , but they travel fastest when they move through a(n) .
Answers: 1
You know the right answer?
Five-gram samples of copper and iron are at room temperature. both samples receive equal amounts of...
Questions



Mathematics, 23.05.2020 03:59

History, 23.05.2020 03:59

Mathematics, 23.05.2020 03:59





History, 23.05.2020 04:00

Mathematics, 23.05.2020 04:00





Mathematics, 23.05.2020 04:00


English, 23.05.2020 04:00

Mathematics, 23.05.2020 04:00

Mathematics, 23.05.2020 04:00

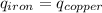
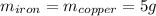 , also
, also