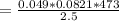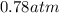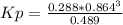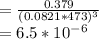Consider the decomposition of the compound C5H6O3 as follows below. C5H6O3(g) → C2H6(g) + 3 CO(g) When a 5.63-g sample of pure C5H6O3(g) was sealed into an otherwise empty 2.50 L flask and heated to 200.°C, the pressure in the flask gradually rose to 1.63 atm and remained at that value. Calculate K for this reaction.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2
You know the right answer?
Consider the decomposition of the compound C5H6O3 as follows below. C5H6O3(g) → C2H6(g) + 3 CO(g) Wh...
Questions

Mathematics, 19.02.2020 21:51





Mathematics, 19.02.2020 21:52

Health, 19.02.2020 21:52

Mathematics, 19.02.2020 21:52

Business, 19.02.2020 21:52




Computers and Technology, 19.02.2020 21:52




Mathematics, 19.02.2020 21:53