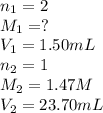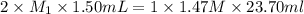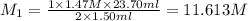Chemistry, 02.03.2020 19:18 coltonsteighner
A 1.50 mL sample of a sulfuric acid solution from an automobile storage battery is titrated with 1.47 M sodium hydroxide solution to a phenolphthalein endpoint, requiring 23.70 mL. What is the molarity of the sulfuric acid solution

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Why should the scientific method be used to answer a question? a. it provides a way to test an idea without any bias. b. it provides a way to test a hypothesis. c. it provides a way to ensure all hypotheses are proven correct. d. it provides a way to quickly turn a hypothesis into a scientific theory.
Answers: 1

Chemistry, 21.06.2019 17:30
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 11:40
Which type of precipitation would most likely form when the surface air temperature is slightly below freezing and the air temperature increases as you move upward away from the ground?
Answers: 2
You know the right answer?
A 1.50 mL sample of a sulfuric acid solution from an automobile storage battery is titrated with 1.4...
Questions






Computers and Technology, 06.07.2019 03:00



Mathematics, 06.07.2019 03:00






Biology, 06.07.2019 03:00


Chemistry, 06.07.2019 03:00


Health, 06.07.2019 03:00

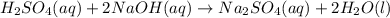
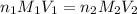
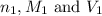 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 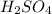
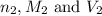 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.