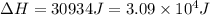Chemistry, 02.03.2020 17:55 shawnr6989
Calculate the amount of heat required to convert 10.0 grams of ice at –20.°C to steam at 120.°C. (Sp. heat of H2O(s) = 2.09 J/g•°C, Sp. heat of H2O(l) = 4.18 J/g•°C, Sp heat of H2O(g) = 2.03 J/g•°C; heat of fus. of H2O(s) = 333 J/g, heat of vap. of H2O(l) = 2260 J/g).

Answers: 3


Another question on Chemistry

Chemistry, 20.06.2019 18:04
If a certain battery supplies 1.0×108 electrons per second to the negative terminal and the battery contains 7.00 moles of electrolytic solution (which means the solution contains 7.00 moles of hso4), what fraction of the solution undergoes a chemical reaction each second
Answers: 1

Chemistry, 21.06.2019 15:50
Nitrogen dioxide reacts with water to form nitric acid and nitrogen monoxide according to the equation: 3no2(g)+h2o(l)→2hno3(l)+no(g) part a suppose that 4.2 mol no2 and 0.50 mol h2o combine and react completely. which reactant is in excess? express your answer as a chemical formula. nothing
Answers: 1


Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1
You know the right answer?
Calculate the amount of heat required to convert 10.0 grams of ice at –20.°C to steam at 120.°C. (Sp...
Questions


Mathematics, 26.02.2021 17:10


Mathematics, 26.02.2021 17:10

Social Studies, 26.02.2021 17:10



Mathematics, 26.02.2021 17:10


Biology, 26.02.2021 17:10



Biology, 26.02.2021 17:10


Biology, 26.02.2021 17:10


Mathematics, 26.02.2021 17:10



Mathematics, 26.02.2021 17:10

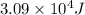
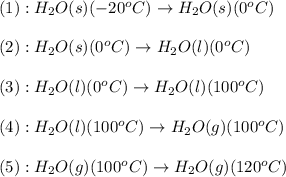
![\Delta H=[m\times c_{p,s}\times (T_{final}-T_{initial})]+m\times \Delta H_{fusion}+[m\times c_{p,l}\times (T_{final}-T_{initial})]+m\times \Delta H_{vap}+[m\times c_{p,g}\times (T_{final}-T_{initial})]](/tpl/images/0530/2584/4a4bb.png)
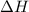 = heat required for the reaction
= heat required for the reaction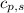 = specific heat of solid water or ice =
= specific heat of solid water or ice = 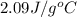
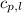 = specific heat of liquid water =
= specific heat of liquid water = 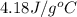
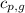 = specific heat of gaseous water =
= specific heat of gaseous water = 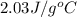
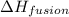 = enthalpy change for fusion =
= enthalpy change for fusion = 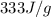
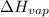 = enthalpy change for vaporization =
= enthalpy change for vaporization = 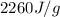
![\Delta H=[10.0g\times 2.09J/g^oC\times (0-(-20))^oC]+10.0g\times 333J/g+[10.0g\times 4.18J/g^oC\times (100-0)^oC]+10.0g\times 2260J/g+[10.0g\times 2.03J/g^oC\times (120-100)^oC]](/tpl/images/0530/2584/295b1.png)