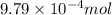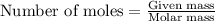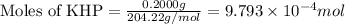Chemistry, 02.03.2020 17:28 janreyes39
As part of Lab 4 you will make and standardize a solution of NaOH(aq). Suppose in the lab you measure the solid NaOH and dissolve it into 100.0 mL of water. You then measure 0.2000 g of KHP (204.22 g/mol) and place it in a clean, dry 100-mL beaker, and then dissolve the KHP in about 25 mL of water and add a couple of drops of phenolphthalein indicator. You titrate this with your NaOH(aq) solution and find that the titration requires 9.78 mL of NaOH(aq). Part 1: How many moles of KHP are in your sample

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1


Chemistry, 22.06.2019 19:50
Which sentence from holes contains an implied personality trait? stanley and his parents had tried to pretend that he was just going away to camp for a while, just like rich kids do. he'd just been in the wrong place at the wrong time. stanley felt somewhat dazed as the guard unlocked his handcuffs and led him off the bus. stanley nodded to show he understood
Answers: 3
You know the right answer?
As part of Lab 4 you will make and standardize a solution of NaOH(aq). Suppose in the lab you measur...
Questions




Social Studies, 27.03.2020 04:32

Computers and Technology, 27.03.2020 04:32


English, 27.03.2020 04:32






Mathematics, 27.03.2020 04:32




Mathematics, 27.03.2020 04:32

Mathematics, 27.03.2020 04:32


History, 27.03.2020 04:33