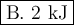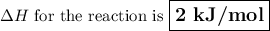Calculate the enthalpy change,,ΔH=∑ Hp - ∑ HR for the following reaction using equations 1, 2 C graphite(s) --> C diamond(s) The following is known. 1. Cgraphite(s) + O2(g) --> CO2(g) ∆H = -394 kJ 2. Cdiamond(s) + O2(g) --> CO2(g) ∆H = -396 kJ A. -763kJ B. 2kJ C. 790kJ D. -2kJ

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Which of the following mining methods disrupts the sea floor?
Answers: 1

Chemistry, 21.06.2019 18:30
Amass of 100.0 g of solute is dissolved in water so that 850. ml of a 0.7500 m solution has been prepared. what is the molar mass of the solute?
Answers: 2

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1
You know the right answer?
Calculate the enthalpy change,,ΔH=∑ Hp - ∑ HR for the following reaction using equations 1, 2 C grap...
Questions

Biology, 16.04.2020 20:00

Biology, 16.04.2020 20:00


Mathematics, 16.04.2020 20:00

Mathematics, 16.04.2020 20:00

History, 16.04.2020 20:01



Mathematics, 16.04.2020 20:01

Mathematics, 16.04.2020 20:01




Social Studies, 16.04.2020 20:01


Physics, 16.04.2020 20:01



History, 16.04.2020 20:01