Below are the reduction half reactions for chemolithoautotrophic nitrification, where ammonia is a source of electrons and energy and oxygen is the terminal electron acceptor. NO2- 6e- -> NH4 (-0.41 volts) O2 4e- -> 2H2O ( 0.82 volts) If you balance and combine the reactions so that 28 molecules of NH4 are oxidized to NO2-, how many molecules of O2 will be reduced to H2O

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Based on the equation and the information in the table, what is the enthalpy of the reaction?
Answers: 2

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 12:30
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1
You know the right answer?
Below are the reduction half reactions for chemolithoautotrophic nitrification, where ammonia is a s...
Questions

History, 02.08.2019 01:30

Computers and Technology, 02.08.2019 01:30



History, 02.08.2019 01:30

Mathematics, 02.08.2019 01:30







Biology, 02.08.2019 01:30

Spanish, 02.08.2019 01:30

Mathematics, 02.08.2019 01:30



Social Studies, 02.08.2019 01:30


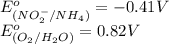
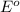 potential will always get reduced and will undergo reduction reaction. Here, oxygen will undergo reduction reaction will get reduced.
potential will always get reduced and will undergo reduction reaction. Here, oxygen will undergo reduction reaction will get reduced. will undergo oxidation reaction and will get oxidized.
will undergo oxidation reaction and will get oxidized.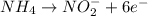 ( × 4)
( × 4)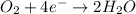 ( × 6)
( × 6)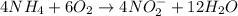
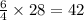 molecules of oxygen gas
molecules of oxygen gas