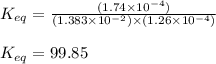Chemistry, 29.02.2020 02:52 eriksprincess13
An aqueous solution was prepared at 21 oC by mixing 7.00 mL 2.00 x 10-2mol L-1Fe3+, 2.00 mL 1.50 x 10-3 mol L-1SCN−, and 1.00 mL water. At equilibrium, the concentration of the product complex, [Fe(SCN)2+]eq was determined to be 1.74 x 10-4mol L-1. What is the value of the equilibrium constant K for the reaction of interest at 21 oC?

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 17:50
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2

Chemistry, 23.06.2019 08:00
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 1
You know the right answer?
An aqueous solution was prepared at 21 oC by mixing 7.00 mL 2.00 x 10-2mol L-1Fe3+, 2.00 mL 1.50 x 1...
Questions


Engineering, 20.04.2021 07:20







Engineering, 20.04.2021 07:20

Mathematics, 20.04.2021 07:20


Mathematics, 20.04.2021 07:20


Mathematics, 20.04.2021 07:20



Mathematics, 20.04.2021 07:20


Biology, 20.04.2021 07:20

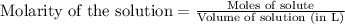 .....(1)
.....(1) ions:
ions: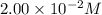
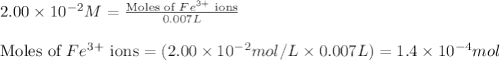
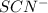 ions:
ions: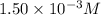
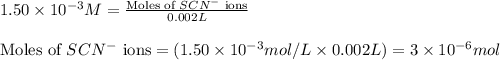
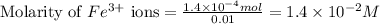
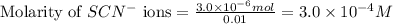
![[FeSCN^{2+}]](/tpl/images/0528/9422/797d4.png) complex follows:
complex follows:![Fe^{2+}+SCN^-\rightleftharpoons [FeSCN^{2+}]](/tpl/images/0528/9422/8a467.png)
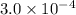
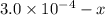 x
x![[FeSCN^{2+}]=1.74\times 10^{-4}M=x](/tpl/images/0528/9422/2c1de.png)
![[Fe^{2+}]\text{ ions}=(1.4\times 10^{-2}-x)=(1.4-0.0174)\times 10^{-3}=1.383\times 10^{-2}M](/tpl/images/0528/9422/f1fb2.png)
![[SCN^{-}]\text{ ions}=(3.0\times 10^{-4}-x)=(3.0-1.74)\times 10^{-4}=1.26\times 10^{-4}M](/tpl/images/0528/9422/6910c.png)
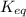 for above equation follows:
for above equation follows:![K_{eq}=\frac{[FeSCN^{2+}]}{[Fe^{3+}][SCN^-]}](/tpl/images/0528/9422/63e7c.png)