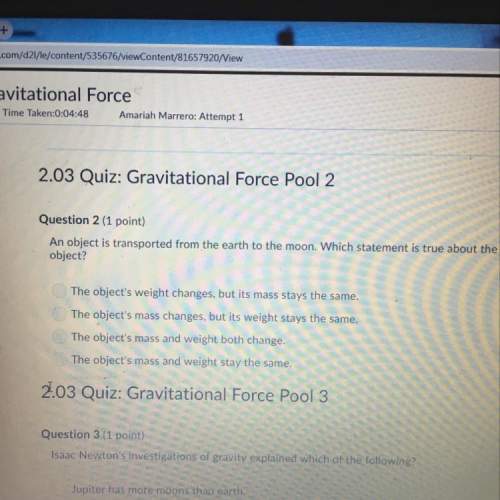
At a particular temperature, K = 3.75 for the following reaction. SO2(g) + NO2(g) equilibrium reaction arrow SO3(g) + NO(g) If all four gases had initial concentrations of 0.550 M, calculate the equilibrium concentrations of the gases. A. M[SO3] WebAssign will check your answer for the correct number of significant figures. B. M [NO] WebAssign will check your answer for the correct number of significant figures. C. M [SO2] WebAssign will check your answer for the correct number of significant figures. D. M [NO2] WebAssign will check your answer for the correct number of significant figures.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Your friend offers to show you an intrusive igneous rock. which of the following would you expect to see?
Answers: 1

Chemistry, 22.06.2019 05:30
Transportation is the largest single source of air pollution in the united states. air pollution can harm the environment and human health. which technology could offer a solution to this problem? mufflers that reduce noise motors that run on electricity tires that improve gas mileage
Answers: 3

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3
You know the right answer?
At a particular temperature, K = 3.75 for the following reaction. SO2(g) + NO2(g) equilibrium reacti...
Questions


English, 19.09.2019 03:00

Biology, 19.09.2019 03:00

Business, 19.09.2019 03:00

Mathematics, 19.09.2019 03:00

Biology, 19.09.2019 03:00

English, 19.09.2019 03:00

Biology, 19.09.2019 03:00

Health, 19.09.2019 03:00


Social Studies, 19.09.2019 03:00

Mathematics, 19.09.2019 03:00


Business, 19.09.2019 03:00

Biology, 19.09.2019 03:00




Biology, 19.09.2019 03:00