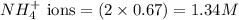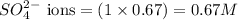Chemistry, 29.02.2020 01:36 kcombest7219
Calculate the concentration of all ions present in each of the following solutions of strong electrolytes. a. 0.0200 mol sodium phosphate in 10.0 mL of solution. 0.300 mol of barium nitrate in 600.0 mL of solution. 1.00 g of potassium chloride in 0.500 L of solution. 132 g of ammonium sulfate in 1.50 L of solution

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
When determining the shape of a molecule, it is important to draw a lewis dot structure first in order to see the total number a. electrons within the moleculeb. bonding and unshared pairs around central atomc. unshared pair within the molecule( i really need it )
Answers: 1

Chemistry, 22.06.2019 01:00
According to the tide table below what time of day will the highest tide occur?
Answers: 1

Chemistry, 22.06.2019 04:30
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Chemistry, 22.06.2019 10:30
Geothermal energy for industrial use is available almost anywhere. a.true b.false
Answers: 2
You know the right answer?
Calculate the concentration of all ions present in each of the following solutions of strong electro...
Questions

Mathematics, 23.03.2021 05:20

Computers and Technology, 23.03.2021 05:20

English, 23.03.2021 05:20


Mathematics, 23.03.2021 05:20

Mathematics, 23.03.2021 05:20






Mathematics, 23.03.2021 05:20






Mathematics, 23.03.2021 05:20

English, 23.03.2021 05:20

Mathematics, 23.03.2021 05:20

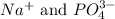 ions in the solution are 6 M and 2 M respectively.
ions in the solution are 6 M and 2 M respectively.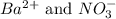 ions in the solution are 0.5 M and 1.0 M respectively.
ions in the solution are 0.5 M and 1.0 M respectively.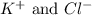 ions in the solution are 0.051 M and 0.051 M respectively.
ions in the solution are 0.051 M and 0.051 M respectively.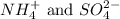 ions in the solution are 1.34 M and 0.67 M respectively.
ions in the solution are 1.34 M and 0.67 M respectively.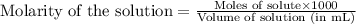 ......(1)
......(1)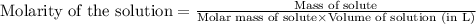 ...(2)
...(2)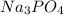
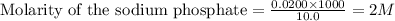
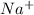 ions and 1 mole of
ions and 1 mole of 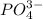 ions
ions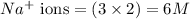
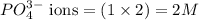
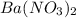
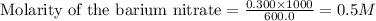
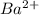 ions and 2 mole of
ions and 2 mole of 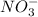 ions
ions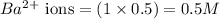
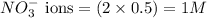
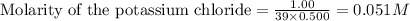
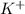 ions and 1 mole of
ions and 1 mole of 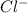 ions
ions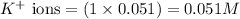
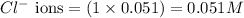
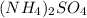
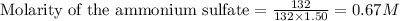
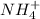 ions and 1 mole of
ions and 1 mole of 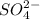 ions
ions