Chemistry, 28.02.2020 22:23 Beast3dgar
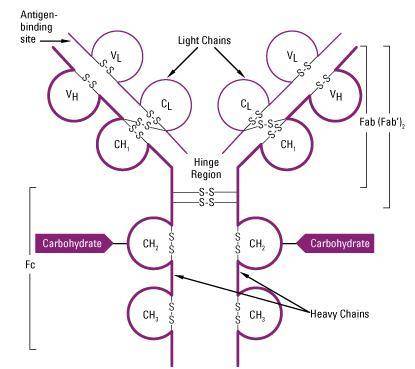
One of the major classes of antibody molecules is immunoglobulin G (IgG). Two heavy chains and two light chains form one IgG molecule. Classify each statement as describing the heavy chains, light chains, or both chains of IgG.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2


Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
You know the right answer?
One of the major classes of antibody molecules is immunoglobulin G (IgG). Two heavy chains and two l...
Questions



Mathematics, 17.10.2019 09:01




Mathematics, 17.10.2019 09:01

Mathematics, 17.10.2019 09:01



Mathematics, 17.10.2019 09:01

English, 17.10.2019 09:01

Computers and Technology, 17.10.2019 09:01



English, 17.10.2019 09:01

Chemistry, 17.10.2019 09:01

Mathematics, 17.10.2019 09:01

Health, 17.10.2019 09:01

Mathematics, 17.10.2019 09:01