
Chemistry, 28.02.2020 19:25 kayleegeise
The equilibrium constant for the reaction below, at a given temperature is 45.6. If the equilibrium concentrations of F2 and BrF3 are 1.24 x 10-1 M and 1.99 x 10-1 M respectively, calculate the equilibrium concentration of Br2. (4)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:50
Choose all that apply. when creating a graph, you should: determine the x- and y- variables label the scale on the x- and y- axes plot the data points draw a line of best fit to represent the data trend
Answers: 1

Chemistry, 22.06.2019 04:30
Why are people not able to scuba dive in the deep part of the ocean
Answers: 2

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3
You know the right answer?
The equilibrium constant for the reaction below, at a given temperature is 45.6. If the equilibrium...
Questions


Mathematics, 05.03.2021 03:10

Mathematics, 05.03.2021 03:10


Mathematics, 05.03.2021 03:10






Mathematics, 05.03.2021 03:10

Chemistry, 05.03.2021 03:10



Mathematics, 05.03.2021 03:10



Chemistry, 05.03.2021 03:10


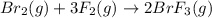
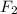 at equilibrium =
at equilibrium = 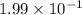
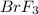 at equilibrium =
at equilibrium = 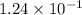
![K_c=\frac{[BrF_3]^2}{[Br_2][F_2]^3}](/tpl/images/0528/1843/a1098.png)
![45.6=\frac{(1.24\times 10^{-1})^2}{[Br_2]\times (1.99\times 10^{-1})^3}](/tpl/images/0528/1843/8cd8d.png)
![[Br_2]=0.0428M](/tpl/images/0528/1843/2a0dc.png)



