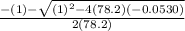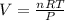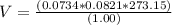Consider the reaction below for which K = 78.2 atm-1. A(g) + B(g) ↔ C(g) Assume that 0.386 mol C(g) is placed in the cylinder represented below. The barometric pressure on the piston (which is assumed to be massless and frictionless) is constant at 1.00 atm. The original volume (before the 0.386 mol C(g) begins to decompose) is 7.29 L. The temperature is fixed and can be determined from the idea gas law. What is the volume in the cylinder at equilibrium?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

Chemistry, 22.06.2019 22:30
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1
You know the right answer?
Consider the reaction below for which K = 78.2 atm-1. A(g) + B(g) ↔ C(g) Assume that 0.386 mol C(g)...
Questions


Mathematics, 25.02.2021 21:50


Mathematics, 25.02.2021 21:50


Mathematics, 25.02.2021 21:50

Mathematics, 25.02.2021 21:50

Mathematics, 25.02.2021 21:50

English, 25.02.2021 21:50


Biology, 25.02.2021 21:50


History, 25.02.2021 21:50



Computers and Technology, 25.02.2021 21:50


Chemistry, 25.02.2021 21:50


Mathematics, 25.02.2021 21:50

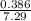
![K = \frac{[C]}{[A][B]}](/tpl/images/0526/7975/63063.png)
![78.2=\frac{[0.0530-x]}{[x][x]}](/tpl/images/0526/7975/8334d.png)
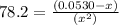
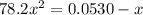
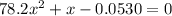
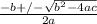
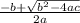 or
or 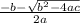
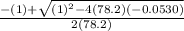 or
or