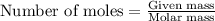Write the balanced equation for the reaction of aqueous Pb ( ClO 3 ) 2 Pb(ClO3)2 with aqueous NaI . NaI. Include phases. chemical equation: What mass of precipitate will form if 1.50 L 1.50 L of highly concentrated Pb ( ClO 3 ) 2 Pb(ClO3)2 is mixed with 0.400 L 0.130 M NaI 0.400 L 0.130 M NaI ? Assume the reaction goes to completion

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Urea, co(nh2)2, is manufactured on a large scale for use in producing urea-formaldehyde plastics and as a fertilizer. what is the maximum mass of urea that can be manufactured from the co2 produced by combustion of 1.00 x 104 grams of co2?
Answers: 1

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3


Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1
You know the right answer?
Write the balanced equation for the reaction of aqueous Pb ( ClO 3 ) 2 Pb(ClO3)2 with aqueous NaI ....
Questions


Biology, 12.12.2020 16:50



English, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50




Mathematics, 12.12.2020 16:50


Mathematics, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50

Spanish, 12.12.2020 16:50



Social Studies, 12.12.2020 16:50


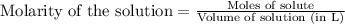
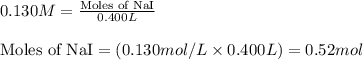
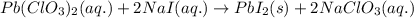
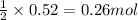 of lead (II) iodide
of lead (II) iodide