Chemistry, 27.02.2020 00:31 dakotakeating4513
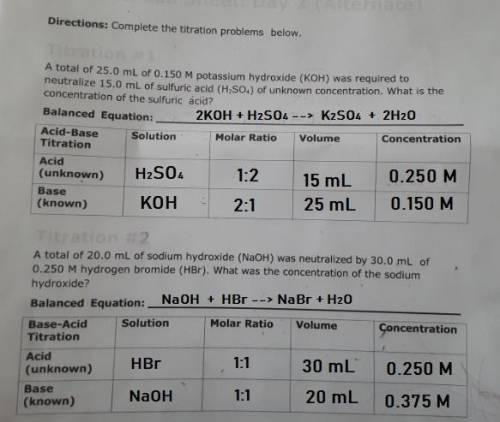
PLEASE HELP! A total of 25.0 mL of 0.150 M potassium hydroxide (KOH) was required to neutralize 15.0 mL of sulfuric acid (H2SO4) of unknown concentration. What is the concentration of the sulfuric acid?
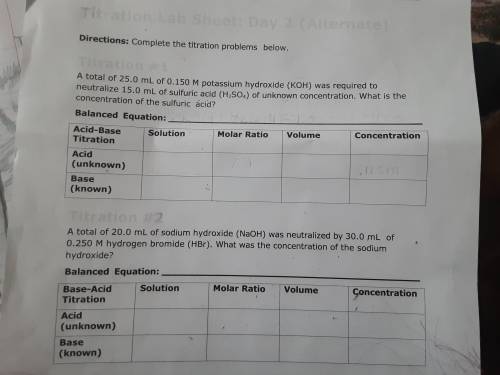

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asmall amount of a solid is added to water. the observation made after fifteen minutes is shown in the figure. which of these solids has been probably added to water? a) oil b) sand c) sugar d) wood chips
Answers: 1

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3


Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2
You know the right answer?
PLEASE HELP! A total of 25.0 mL of 0.150 M potassium hydroxide (KOH) was required to neutralize 15.0...
Questions


History, 18.12.2020 01:00

Mathematics, 18.12.2020 01:00

Social Studies, 18.12.2020 01:00

Mathematics, 18.12.2020 01:00





Mathematics, 18.12.2020 01:00


Health, 18.12.2020 01:00


English, 18.12.2020 01:00




Mathematics, 18.12.2020 01:00

Computers and Technology, 18.12.2020 01:00

Biology, 18.12.2020 01:00