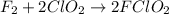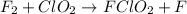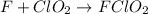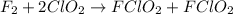Chemistry, 26.02.2020 04:19 kaylabay1997
A mechanism for the gas phase reaction of fluorine with chlorine dioxide that is consistent with the observed rate law is:
step 1 slow: F2 + ClO2 > FClO2 + F
step 2 fast: F > ClO2 + FClO2
What is the equation for the overall reaction?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1


Chemistry, 22.06.2019 19:00
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
A mechanism for the gas phase reaction of fluorine with chlorine dioxide that is consistent with the...
Questions


Mathematics, 26.10.2020 05:50

Mathematics, 26.10.2020 05:50

Arts, 26.10.2020 05:50

Geography, 26.10.2020 05:50

Mathematics, 26.10.2020 05:50




English, 26.10.2020 05:50

Mathematics, 26.10.2020 05:50

English, 26.10.2020 05:50


English, 26.10.2020 05:50

Mathematics, 26.10.2020 05:50

Mathematics, 26.10.2020 05:50


Arts, 26.10.2020 05:50


English, 26.10.2020 05:50