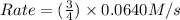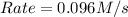Chemistry, 26.02.2020 03:09 abdullahs4639
The reaction A + B ⟶ C + D rate = k [ A ] [ B ] 2 A+B⟶C+Drate=k[A][B]2 has an initial rate of 0.0640 M / s. 0.0640 M/s. What will the initial rate be if [ A ] [A] is halved and [ B ] [B] is tripled?What will the initial rate be if [A] is tripled and [B] is halved? answer M/s

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1
You know the right answer?
The reaction A + B ⟶ C + D rate = k [ A ] [ B ] 2 A+B⟶C+Drate=k[A][B]2 has an initial rate of 0.0640...
Questions

World Languages, 03.10.2019 07:30



Chemistry, 03.10.2019 07:30

History, 03.10.2019 07:30





Mathematics, 03.10.2019 07:30

Mathematics, 03.10.2019 07:30

Mathematics, 03.10.2019 07:30

Chemistry, 03.10.2019 07:30

Spanish, 03.10.2019 07:30


Biology, 03.10.2019 07:30

Mathematics, 03.10.2019 07:30



Mathematics, 03.10.2019 07:30

![Rate=k[A][B]^2](/tpl/images/0524/3846/d8f90.png)
![Rate=k\times (\frac{[A]}{2})\times (3\times [B])^2](/tpl/images/0524/3846/31c7e.png)
![Rate=k\times (\frac{[A]}{2})\times 9\times [B]^2](/tpl/images/0524/3846/e768d.png)
![Rate=k\times (\frac{9}{2})\times [A]\times [B]^2](/tpl/images/0524/3846/4e890.png)
![k[A][B]^2](/tpl/images/0524/3846/8ef61.png) = 0.0640 M/s
= 0.0640 M/s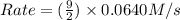
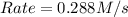
![Rate=k\times (\frac{[B]}{2})^2\times (3\times [A])](/tpl/images/0524/3846/fb264.png)
![Rate=k\times (\frac{[B]^2}{4})\times 3\times [A]](/tpl/images/0524/3846/c4659.png)
![Rate=k\times (\frac{3}{4})\times [A]\times [B]^2](/tpl/images/0524/3846/8107a.png)