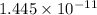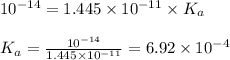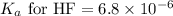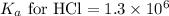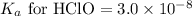Chemistry, 26.02.2020 02:26 ashleypere99
An unknown salt is either NaF, NaCl, or NaOCl. When 0.050 mol of the salt is dissolved in water to form 0.500 L of solution, the pH of the solution is 8.08. Part A What is the identity of the salt?

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 06:00
Jenny wants to test the electrical conductivity of two substances dissolved in water. she is preparing the containers for the experiment. which factor is most important for her to control?
Answers: 1

Chemistry, 23.06.2019 13:30
Determine the rate law, including the values of the orders and rate law constant, for the following reaction using the experimental data provided. a + b yields products trial [a] [b] rate 1 0.30 m 0.25 m 1.2 × 10-2 m/min 2 0.30 m 0.50 m 4.8 × 10-2 m/min 3 0.60 m 0.50 m 9.6 × 10-2 m/min
Answers: 1

Chemistry, 23.06.2019 14:00
Beaker a contains 100 ml of 1.5 mammonia. beaker b contains 100 ml of 0.50m ammonia. how does the solution in beaker a compare to the solution in beaker b?
Answers: 2

Chemistry, 23.06.2019 15:00
What do we call the rows on the periodic table? a. periodb. familyc. groupd. metals
Answers: 1
You know the right answer?
An unknown salt is either NaF, NaCl, or NaOCl. When 0.050 mol of the salt is dissolved in water to f...
Questions



Social Studies, 24.03.2020 06:25






Mathematics, 24.03.2020 06:26


History, 24.03.2020 06:26


English, 24.03.2020 06:26


History, 24.03.2020 06:26

Mathematics, 24.03.2020 06:26


Mathematics, 24.03.2020 06:26

Social Studies, 24.03.2020 06:26

Mathematics, 24.03.2020 06:26

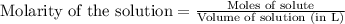

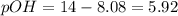
![pOH=-\log[OH^-]](/tpl/images/0524/3194/fe336.png)
![5.92=-\log[OH^-]](/tpl/images/0524/3194/4c04e.png)
![[OH^-]=10^{-5.92}=1.202\times 10^{-6}M](/tpl/images/0524/3194/90070.png)
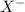 ions follows:
ions follows: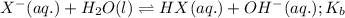
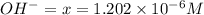
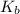 for above equation follows:
for above equation follows:![K_b=\frac{[OH^-][HX]}{[X^-]}](/tpl/images/0524/3194/2b2a0.png)
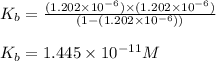
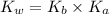
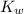 = Ionic product of water =
= Ionic product of water = 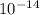
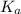 = Acid dissociation constant
= Acid dissociation constant