
Chemistry, 26.02.2020 02:23 jcrowley9362
Calcium carbide, CaC2, used to produce acetylene, C2H2, is prepared by heating calcium oxide and carbon to high temperature. Carbon monoxide gas is formed as a by-product. a. Write down the balanced equation for this process. b. If a mixture contains 1.15 kg of each reactant, how many grams of calcium carbide can be prepared?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1

Chemistry, 23.06.2019 10:50
Gene expression control that occurs during the generation of rna is a. controlled at transcription b. control before transcription c. controlled after transcription d. controlled after translation
Answers: 3

Chemistry, 23.06.2019 14:30
Recognizing the properties of water water has a "bent" geometry. which explanation does not explain why? o water's oxygen has unbonded electron pairs that repel each other. water can form hydrogen bonds. electrons are evenly distributed in the water molecule. do ne
Answers: 3
You know the right answer?
Calcium carbide, CaC2, used to produce acetylene, C2H2, is prepared by heating calcium oxide and car...
Questions

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Physics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

English, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

Mathematics, 10.09.2020 20:01

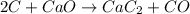
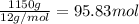
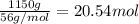
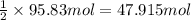 of CaO.
of CaO.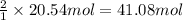 of carbon
of carbon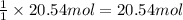 of calcium carbide
of calcium carbide

