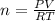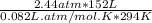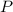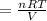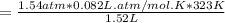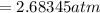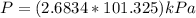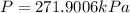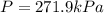On a summer day, you take a road trip through Moses Lake, WA, in a sports car. You start out at a temperature of 21°C in the morning, but the temperature in Moses Lake will reach a peak of 55°C. Each tire on your car holds 15.2 L of nitrogen gas at a starting pressure of 247 kPa. The tires will burst if the internal pressure exceeds 270 kPa.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:10
Amonoprotic acid is an acid that donates a single proton to the solution. suppose you have 0.140 g of a monoprotic acid dissolved in 35.0 ml of water. this solution is then neutralized with 14.5 ml of 0.110 m naoh. what is the molar mass of the acid?
Answers: 1

Chemistry, 21.06.2019 16:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change. when the temperature in a room increases from 25°c to 33°c, changes from a solid to a liquid. in a lab, methane and nitrogen are cooled from -170°c to -200°c. the methane freezes and the nitrogen . when gold is heated to 2,856°c it changes from a liquid to a .
Answers: 2

Chemistry, 22.06.2019 05:30
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3
You know the right answer?
On a summer day, you take a road trip through Moses Lake, WA, in a sports car. You start out at a te...
Questions

Mathematics, 02.01.2020 06:31



Chemistry, 02.01.2020 06:31

Mathematics, 02.01.2020 06:31



Chemistry, 02.01.2020 06:31

Mathematics, 02.01.2020 06:31

Mathematics, 02.01.2020 06:31

Mathematics, 02.01.2020 06:31

Mathematics, 02.01.2020 06:31


History, 02.01.2020 06:31

Geography, 02.01.2020 06:31

Spanish, 02.01.2020 06:31




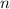
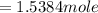
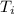 ) = 21°C = (21 +273)K
) = 21°C = (21 +273)K