
The reaction below is carried out at a different temperature at which Kc=0.055. This time, however, the reaction mixture starts with only the product, [NO]=0.0100M, and no reactants. Find the equilibrium concentrations of N2, O2, and NO at equilibrium. N2(g)+O2(g)⇌2NO(g)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

You know the right answer?
The reaction below is carried out at a different temperature at which Kc=0.055. This time, however,...
Questions

Biology, 05.10.2019 06:00

Mathematics, 05.10.2019 06:00

Health, 05.10.2019 06:00

Physics, 05.10.2019 06:00



Mathematics, 05.10.2019 06:00

Mathematics, 05.10.2019 06:00

French, 05.10.2019 06:00

Mathematics, 05.10.2019 06:00


Mathematics, 05.10.2019 06:00

Chemistry, 05.10.2019 06:00


Mathematics, 05.10.2019 06:00

History, 05.10.2019 06:00



Biology, 05.10.2019 06:00

Mathematics, 05.10.2019 06:00

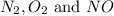 at equilibrium is, 0.0045 M, 0.0045 M and 0.001 M respectively.
at equilibrium is, 0.0045 M, 0.0045 M and 0.001 M respectively.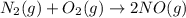
![K=\frac{[NO]^2}{[N_2][O_2]}](/tpl/images/0523/5256/ef70d.png)
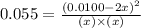
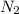 at equilibrium = x = 0.0045 M
at equilibrium = x = 0.0045 M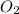 at equilibrium = x = 0.0045 M
at equilibrium = x = 0.0045 M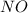 at equilibrium = (0.0100-2x) = (0.0100-2(0.0045)) = 0.001 M
at equilibrium = (0.0100-2x) = (0.0100-2(0.0045)) = 0.001 M

