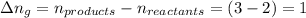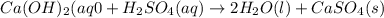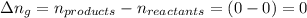Chemistry, 25.02.2020 03:04 DaFuzzyDude
The enthalpy change, ΔH, for a reaction at constant pressure is defined as: ΔH = ΔE + PΔV. For which of the following reactions will ΔH be approximately equal to ΔE? Select all that apply. Group of answer choices 2 NO2(g) -> N2(g) + 2 O2(g)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:10
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 18:00
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1
You know the right answer?
The enthalpy change, ΔH, for a reaction at constant pressure is defined as: ΔH = ΔE + PΔV. For which...
Questions


Mathematics, 27.01.2020 17:31


History, 27.01.2020 17:31


Mathematics, 27.01.2020 17:31




History, 27.01.2020 17:31

History, 27.01.2020 17:31



Physics, 27.01.2020 17:31





History, 27.01.2020 17:31

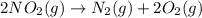
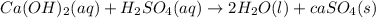
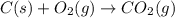
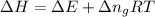 as
as 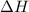 = enthalpy change
= enthalpy change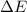 = internal energy change
= internal energy change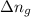 = change in number of moles of gas particles =
= change in number of moles of gas particles =