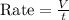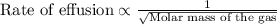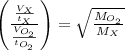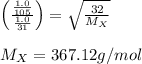A gas of unknown molecular mass was allowed to effuse through a small opening under constant-pressure conditions. It required 105 s for 1.0 L of the gas to effuse. Under identical experimental conditions it required 31 s for 1.0 L of O2 gas to effuse. Calculate the molar mass of the unknown gas.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Sarah wants to know where in her garden chamomile would grow the best. she thinks chamomile will grow best in the corner of the garden that gets the most sunlight. to test her hypothesis, she decides to plant several groups of chamomile in her garden as an experiment. which of the following variables will sarah need to measure to know which group of plants grew best? a. the location of the plants b. the type of plants c. the height of the plants d. the amount of water she gives the plants
Answers: 1

Chemistry, 22.06.2019 10:30
When the speed of the bottle is 2 m/s, the average maximum height of the beanbag is m.
Answers: 2

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1
You know the right answer?
A gas of unknown molecular mass was allowed to effuse through a small opening under constant-pressur...
Questions


Mathematics, 18.10.2020 15:01


History, 18.10.2020 15:01

Mathematics, 18.10.2020 15:01

Social Studies, 18.10.2020 15:01


Social Studies, 18.10.2020 15:01



Chemistry, 18.10.2020 15:01

History, 18.10.2020 15:01

English, 18.10.2020 15:01

English, 18.10.2020 15:01

Mathematics, 18.10.2020 15:01


Physics, 18.10.2020 15:01