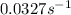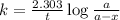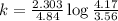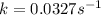Chemistry, 24.02.2020 23:41 davisbrittany5784
Consider the following first-order reaction: A → B. The concentration of A at the start of the reaction is 4.17 M and after 4.84 s is 3.56 M. (a) Using the integrated rate law for a first-order reaction, calculate the value of the rate constant.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:00
3. if the dartboard below is used to model an atom, which subatomic particles would be located at z?
Answers: 2

Chemistry, 22.06.2019 08:00
Will give ! what are the advantages and disadvantages of nuclear power? check all that apply. one advantage of nuclear energy is that it does not produce carbon dioxide emissions. storage of nuclear waste is a short-term problem associated with nuclear energy. the problem with uranium mining is that a large quantity of uranium must be extracted to meet energy needs because the energy release from uranium fission is so low. safe operation of a nuclear power plant can be jeopardized by a human mistake.
Answers: 1

Chemistry, 22.06.2019 18:30
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1

You know the right answer?
Consider the following first-order reaction: A → B. The concentration of A at the start of the react...
Questions



Mathematics, 23.04.2021 23:40


Arts, 23.04.2021 23:40

Mathematics, 23.04.2021 23:40

Mathematics, 23.04.2021 23:40

Mathematics, 23.04.2021 23:40

Mathematics, 23.04.2021 23:40




Mathematics, 23.04.2021 23:40

Mathematics, 23.04.2021 23:40


Mathematics, 23.04.2021 23:40

Social Studies, 23.04.2021 23:40

Physics, 23.04.2021 23:40

Mathematics, 23.04.2021 23:40