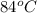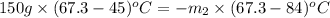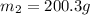Chemistry, 24.02.2020 23:40 korirosekc
A 150 g sample of water (initially at 45 C) is mixed with an unknown mass of water (initially at 84 C). the final temperature of the system is 67.3 C. Determine the mass of water

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
This is a characteristic of the elements in the periodic table that shows a pattern. it may increase or decrease across or down the table.
Answers: 1

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1
You know the right answer?
A 150 g sample of water (initially at 45 C) is mixed with an unknown mass of water (initially at 84...
Questions


Mathematics, 24.07.2019 08:50


Mathematics, 24.07.2019 08:50




English, 24.07.2019 08:50

History, 24.07.2019 08:50

Geography, 24.07.2019 08:50

History, 24.07.2019 08:50

Social Studies, 24.07.2019 08:50



Biology, 24.07.2019 08:50


English, 24.07.2019 08:50


History, 24.07.2019 08:50

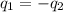
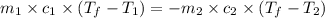
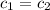 = specific heat water
= specific heat water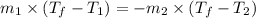
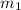 = mass of water = 150 g
= mass of water = 150 g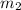 = mass of unknown water = ?
= mass of unknown water = ?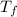 = final temperature of mixture =
= final temperature of mixture = 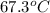
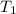 = initial temperature of water =
= initial temperature of water = 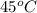
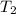 = initial temperature of unknown water =
= initial temperature of unknown water =