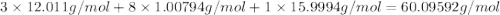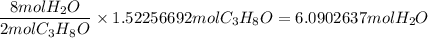Chemistry, 24.02.2020 07:43 bridgettebach
When 91.5 g of isopropyl alcohol which has an empirical formula of C3H8O is burned in excess oxygen gas, how many grams of H2O are formed? MWC = 12.011 g/mol, MWH = 1.00794 g/mol, and MWO = 15.9994 g/mol.
1. 47.9229
2. 84.1255
3. 52.2617
4. 49.8948
5. 86.9152
6. 119.705
7. 88.0758
8. 76.2076
9. 62.9663
10. 109.729

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is a scientific theory? a. a scientist's guess about how something works b. the results of an experiment obtained using the scientific method c. a proven fact that will never change d. an idea that is backed by data from many sources
Answers: 2

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 16:00
Is a measure of the resistance to flow. a high liquid has a high resistance to flow and flows slowly. the ancients thought everything in the world was made of 4 we now know that there are 94 naturally occurring and scientists have created another 24 i am certain they will create even more. honey flows slowly because it has a high to flowing. a can be separated by physical means because it contains more than one pure substance and 2 pure substances are not chemically bonded to each other. a cannot be separated by physical means. all matter is made up of all elements are with the same number of protons. if it is just a single or many bonded together, if all of them have the same number of protons, it is an element. in a piece of pure iron metal, all the are joined together, that piece of iron metal is called elemental iron. a single of iron is called elemental iron. a mixture has differences from place to place. we might need a microscope to see them or they might be obvious to the unaided eye. there are surfaces separating it into different phases. a mixture is the same everywhere. it is uniform. there are no surfaces separating it into different phases. if different kinds of atoms (different elements) are bonded together by their electrons, it is called a there are physical means of to isolate the different pure substances in a mixture and there are chemical means of to isolate the different elements in a compound. 1. element 2. compound 3. mixture 4. heterogeneous 5. homogeneous 6. pure substance 7. atoms 8. separation 9. viscosity 10. resistance
Answers: 2

Chemistry, 22.06.2019 18:30
When a device is used in a circuit in which the voltage is 81 v the current flowing through the device is 3 a what is the resistance of the device
Answers: 2
You know the right answer?
When 91.5 g of isopropyl alcohol which has an empirical formula of C3H8O is burned in excess oxygen...
Questions


Social Studies, 22.01.2021 01:00

History, 22.01.2021 01:00


Chemistry, 22.01.2021 01:00






Social Studies, 22.01.2021 01:00







History, 22.01.2021 01:00

Biology, 22.01.2021 01:00

Mathematics, 22.01.2021 01:00

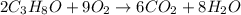
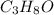 produce 8 moles of
produce 8 moles of 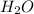 .
.