
Chemistry, 22.02.2020 05:44 marykayy0218own5cp
Hemoglobin molecules in blood bind oxygen and carry it to cells, where it takes part in metabolism. The binding of oxygen hemoglobin(aq) + O2(aq) 88n hemoglobin O2(aq) is rst order in hemoglobin and rst order in dissolved oxygen, with a rate constant of 4 × 107 L mol 1 s 1. Calcu- late the initial rate at which oxygen will be bound to hemo- globin if the concentration of hemoglobin is 2 × 10 9 M and that of oxygen is 5 × 10 5 M.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:30
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1

Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1


Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
You know the right answer?
Hemoglobin molecules in blood bind oxygen and carry it to cells, where it takes part in metabolism....
Questions





Biology, 14.02.2020 18:21

Mathematics, 14.02.2020 18:21


Computers and Technology, 14.02.2020 18:21

Mathematics, 14.02.2020 18:21




English, 14.02.2020 18:22





Mathematics, 14.02.2020 18:22


 +
+ 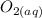 --------->
---------> 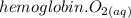
![[hemoglobin_{(aq)}][O_{2(aq)}]](/tpl/images/0520/2072/8993a.png)


