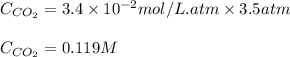Chemistry, 21.02.2020 03:00 Brennen435
The partial pressure of carbon dioxide in a gas mixture is 3.5 atm. What will be the solubility (in M) of carbon dioxide gas when Henry's Law constant for carbon dioxide is 3.4 x 10-2M/atm

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 22.06.2019 04:30
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 14:30
Which of the following is not one of the steps in the scientific method a. hypothesize b. summarize c. analyze d. familiarize
Answers: 3

Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1
You know the right answer?
The partial pressure of carbon dioxide in a gas mixture is 3.5 atm. What will be the solubility (in...
Questions

History, 05.02.2021 22:20

History, 05.02.2021 22:20



Arts, 05.02.2021 22:20


Mathematics, 05.02.2021 22:20

Advanced Placement (AP), 05.02.2021 22:20







English, 05.02.2021 22:20

Mathematics, 05.02.2021 22:20




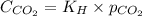
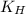 = Henry's constant =
= Henry's constant = 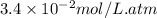
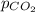 = partial pressure of carbon dioxide gas = 3.5 atm
= partial pressure of carbon dioxide gas = 3.5 atm