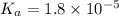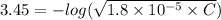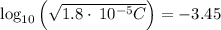Chemistry, 18.02.2020 04:52 Giovanni0312
A solution of acetic acid has a pH of 3.45. What is the concentration of acetic acid in this solution? Ka for CH3COOH is 1.8×10-5.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2


Chemistry, 23.06.2019 07:30
In the diagram here that represents the reaction, which reactant, a or b, is the limiting reagent?
Answers: 1
You know the right answer?
A solution of acetic acid has a pH of 3.45. What is the concentration of acetic acid in this solutio...
Questions

Mathematics, 23.08.2019 17:30


Social Studies, 23.08.2019 17:30



Mathematics, 23.08.2019 17:30










Mathematics, 23.08.2019 17:30

History, 23.08.2019 17:30


Mathematics, 23.08.2019 17:30

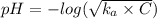
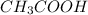 ,
,