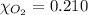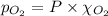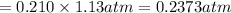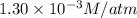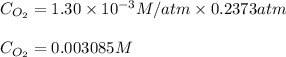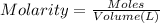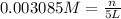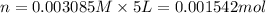Chemistry, 18.02.2020 03:26 netflixacc0107
Calculate the mass of oxygen gas (O2) dissolved in a 5.00 L bucket of water exposed to a pressure of 1.13 atm of air. Assume the mole fraction of oxygen in air to be 0.210 and the Henry's law constant for air in water at this temperature to be 1.30 × 10-3 M/atm. Report your answer in mg.

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3
You know the right answer?
Calculate the mass of oxygen gas (O2) dissolved in a 5.00 L bucket of water exposed to a pressure of...
Questions

Biology, 27.07.2019 11:00

Mathematics, 27.07.2019 11:00


English, 27.07.2019 11:00

History, 27.07.2019 11:00


Social Studies, 27.07.2019 11:00

Mathematics, 27.07.2019 11:00


Mathematics, 27.07.2019 11:00


Mathematics, 27.07.2019 11:00


Biology, 27.07.2019 11:00

Business, 27.07.2019 11:00

History, 27.07.2019 11:00




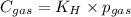
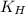 = Henry's constant =
= Henry's constant = 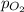 = partial pressure of oxygen
= partial pressure of oxygen