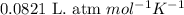Chemistry, 18.02.2020 01:59 MariaGuerra
You analyze a sample of unknown metal as you would in this experiment. You measure the volume of H2(g) generated to be 71.85 mL and the water temperature to be 20.0°C. You calculate PH2 to be 0.781 atm. Use the ideal gas law to calculate the number of moles of hydrogen gas generated.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:50
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1


Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1
You know the right answer?
You analyze a sample of unknown metal as you would in this experiment. You measure the volume of H2(...
Questions


History, 02.04.2020 01:07

History, 02.04.2020 01:07

Mathematics, 02.04.2020 01:07

Mathematics, 02.04.2020 01:07

Mathematics, 02.04.2020 01:07


Mathematics, 02.04.2020 01:07

Mathematics, 02.04.2020 01:07

Mathematics, 02.04.2020 01:07



Mathematics, 02.04.2020 01:07







English, 02.04.2020 01:08

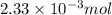
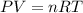
![20^oC=[20+273]K=293K](/tpl/images/0513/7044/3b5d4.png)