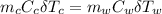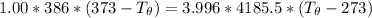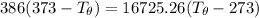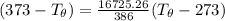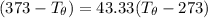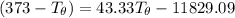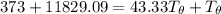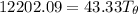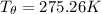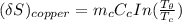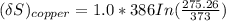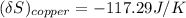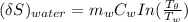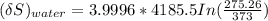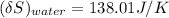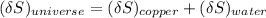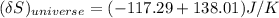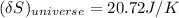Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3

Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1

Chemistry, 23.06.2019 02:00
The plant food contains nh4)3po4 what tests would you run to verify the presence of the nh4 ion and the po4 ion
Answers: 2
You know the right answer?
A 1.00-kg block of copper at 100°C is placed in an in- sulated calorimeter of negligible heat capaci...
Questions


Chemistry, 16.06.2021 04:30

Mathematics, 16.06.2021 04:30

English, 16.06.2021 04:30



Mathematics, 16.06.2021 04:30

Mathematics, 16.06.2021 04:30



Mathematics, 16.06.2021 04:30

Chemistry, 16.06.2021 04:30

History, 16.06.2021 04:30


Mathematics, 16.06.2021 04:30

English, 16.06.2021 04:30




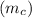 = 1.00 kg
= 1.00 kg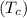 = 100°C
= 100°C 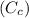 = 386 J/kg.K
= 386 J/kg.K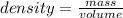
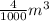
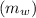 = 3.9996 kg
= 3.9996 kg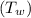 at 0.0°C = 273 K
at 0.0°C = 273 K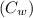 = 4185.5 J/kg.K
= 4185.5 J/kg.K