
Chemistry, 14.02.2020 18:27 kylucienne
The popularity of orange juice, especially as a breakfast drink, makes it an important factor in the economy of orange-growing regions. Marketed juice has either gone through a process in which it was concentrated, or it may be a not-from-concentrate juice. Frozen concentrated juice is reconstituted before consumption. Although concentrated juices are less popular in the United States than at one time, they still have a major segment of the market. The approaches to concentrating orange juice include evaporation, freeze concentration, and reverse osmosis. Here we examine the evaporation process by focusing only on two constituents in the juice: solids and water. Fresh orange juice contains approximately 10.0 wt% solids (sugar, citric acid and other indigenous ingredients) and frozen concentrate contains approximately 42.0 wt% solids. The frozen concentrate is obtained by evaporating water from the fresh juice to produce a mixture that is approximately 57.0 wt% solids. However, so that the flavor of the concentrate will closely approximate that of fresh juice, the concentrate from the evaporator is blended with fresh orange juice (and other minor additives) to produce a final concentrate that is approximately 42.0 wt% solids. Assume a basis of 300.0 kg of fresh juice fed to the process.
Calculate the amount of product (42% concentrate) produced per 100kg fresh juice fed to theprocess and the fraction of the feed that bypasses the evaporator.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 3

Chemistry, 22.06.2019 20:30
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 05:30
How many moles are in 1.26*10^24 particles in significant figures
Answers: 2
You know the right answer?
The popularity of orange juice, especially as a breakfast drink, makes it an important factor in the...
Questions


Mathematics, 01.07.2020 15:01

Chemistry, 01.07.2020 15:01


Biology, 01.07.2020 15:01


Spanish, 01.07.2020 15:01











Mathematics, 01.07.2020 15:01

English, 01.07.2020 15:01

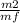 =
= 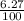 = 0.0627
= 0.0627

